Case Study - FDA - Adverse Event Reporting System
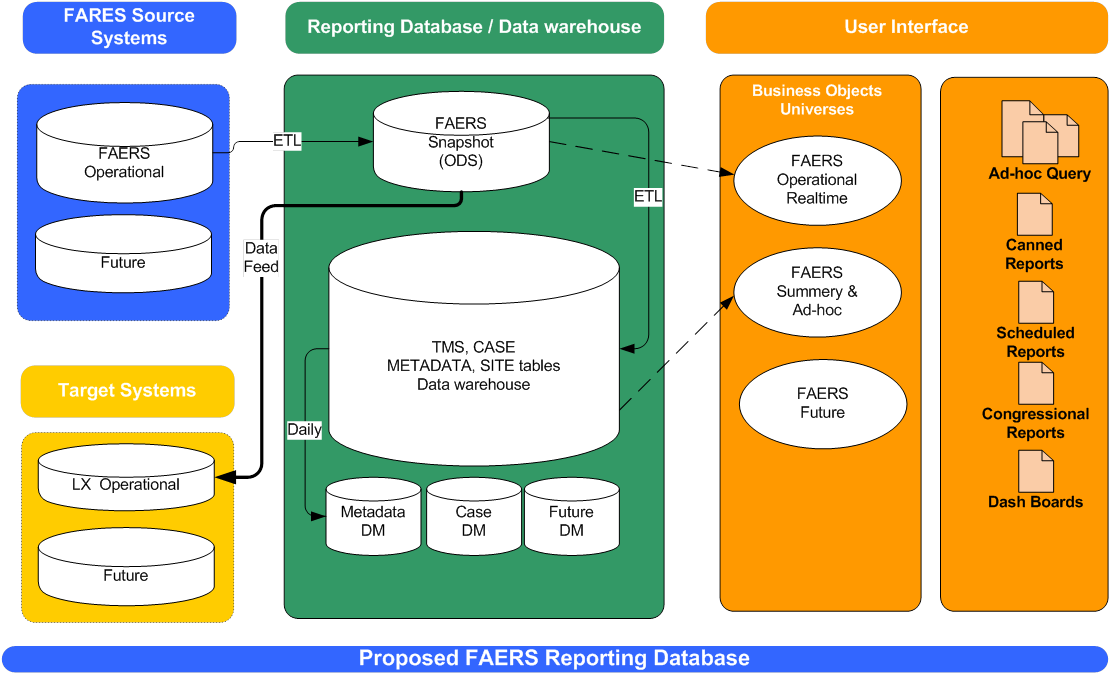
In early 2011, the Office of Information Management (OIM) determined a business intelligence tool was necessary to meet CDER's requirement for complex and robust ad hoc querying, analyzing and reporting functionality for FAERS. Business intelligence tools typically read data stored in warehouses, data marts or replicated data tables. Since FDA had previously selected Business Objects software as an enterprise business intelligence tool, the FAERS Business Intelligence Solution (FBIS) will use Business Objects software for the majority of querying and reporting for FAERS. FBIS will be implemented in two phases. Phase I, which is part of FAERS Release 2.2 (scheduled for delivery in early 2012), will work off a complete replication of the FAERS operational database. FBIS will allow users to perform complex queries, multi-select products and events, drill down to the case level, export case data to Excel-like formats, and produce graphs. For Phase 2 (implementation post Release 2.2), business and OIM will collaborate to enhance FBIS to meet business requirements for specific data provisioning needs that may require transforming or denormalizing the data.